4 Common Challenges in Clinical Trials – How Are You Addressing Them?

We’ve talked at length about the challenges in hospitals, health systems, telehealth, and other aspects of the U.S. healthcare system. Today, let’s take a step back and look at challenges in clinical trials – invaluable studies without which there would be no advances in healthcare.

Preventing professional patients is an overlooked issue in clinical trials.
Clinical trials in a nutshell
Clinical trials have been around since ancient times. In fact, the first clinical trial can be traced back to biblical times – in 500 B.C.! Since then, there have been many changes and updates to how these trials are carried out as well as standards that have to be followed to ensure they are ethical.
Clinical trials are conducted to develop drugs, devices, or procedures that can improve human health. These trials mainly help identify whether the drugs are safe, have any side effects, and can be used by the general population.
While clinical trials used to be a very niche topic, its popularity skyrocketed due to the pandemic. Since the novel virus hit the world, virtually everyone has been asking the same question – when will there be a vaccine? Unfortunately, everyone skips over the challenges in clinical trials and they aren’t discussed as much as the vaccine itself. However, that’s exactly what we’ll look at today – some common challenges faced by organizations to carry out these complex, expensive trials.
4 common challenges in clinical trials
Clinical trials are not at all easy – they’re quite complex, lengthy, and are required to follow several rules and regulations to ensure compliance with different standards. Before starting a clinical trial and while running it, organizations face a number of challenges.
Slower patient recruitment than required
One of the biggest challenges is faced just before starting clinical trials, and it’s all about patient recruitment. Moreover, this is the stage that often determines whether the clinical trial will successfully commence or not as around 80% of these studies are either halted or even closed due to low patient recruitment. One of the reasons for this is the specific requirements of the trial. For instance, the trial requires patients to have a particular condition, but patients have other conditions as well which might lead to complications – finding the right patients proves to be quite a challenge indeed.

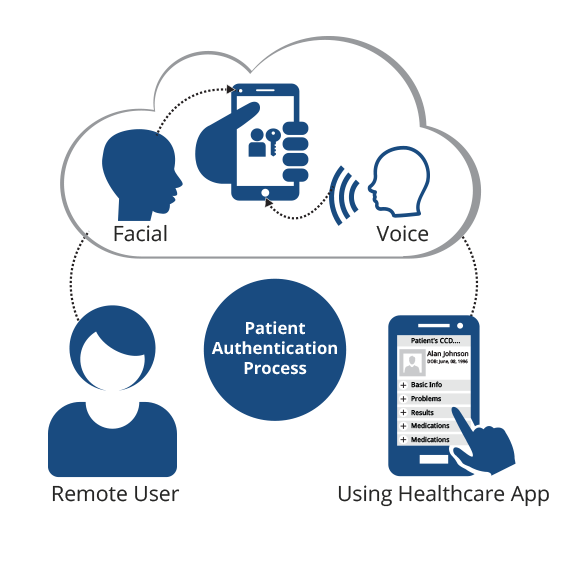
RightPatient addresses one of the overlooked clinical trial challenges – preventing professional patients.
One way many sponsors deal with this is by setting realistic expectations and assuming the fact that not all of their sites will attract the desired number of patients. Another way sponsors tackle the challenge is by outsourcing recruitment since there are several organizations available that specialize in doing so.
Compliance with several rules and regulations
Overcoming the patient recruitment challenge is just the beginning as there’s another herculean task. Ensuring compliance with the rules and regulations has to be ensured throughout the duration of the trial, as it cannot be completed successfully unless the trial conforms to the rules and regulations to a T.
Since trials are full of complex activities, involve human subjects, untested drugs, devices, and procedures, ensuring compliance becomes mandatory to ensure patient safety as well as maintaining ethics of the trials. Usually, institutional review boards (IRBs) have to approve the processes, and that can be time-consuming as there are many factors to consider, which often create a bottleneck.
Managing multiple sites
While clinical trials usually occur over multiple sites to involve diverse patients, more sites mean more vendors, procedures, diverse compliance requirements, and coordination efforts, leading to more complexities down the line.
Creating a robust policy of how to deal with multiple sites and coordinate with one another is one of the only ways forward. Moreover, ensuring accurate patient records and properly managing patients is an absolute must to prevent mix-ups, inconsistencies, and ensure compliance.
Preventing professional patients
Professional patients, or duplicate subjects, are an overlooked but significant issue that hampers clinical trials and threatens the integrity of the trials they participate in. Professional patients sign up for multiple trials either simultaneously or consecutively, which seriously affects the clinical trials. They can even lead to skewing overall data which might render promising drugs unusable since the data shows that the drugs are not effective enough.
Not only do professional patients harm themselves, but they cause billions of dollars in losses or delays in approvals from pharmaceutical regulatory agencies. As a result, preventing professional patients is a must – something which RightPatient can help with.
RightPatient is a leading touchless biometric patient identification platform that ensures patients are accurately identified across the care continuum – several hospitals and health systems are already using it. Fortunately, it can prevent professional patients in clinical trials as well by validating that the patients’ identities and checking whether they are currently participating in other active trials or not. Not only can RightPatient help reduce significant losses, but it also helps protect the integrity of clinical trials – reducing errors and preventing professional patients from participating.










This blog is very helpful and informative for this particular topic. I appreciate your effort that has been taken to write this blog for us.
Great Read. Thank you for this helpful information
great article..
Thank you for providing this information about the challenges of conducting a clinical trial, Matt. I find it interesting that compliance with the rules and regulations to a T can be challenging. I’d imagine that it would be much easier to manage a trial overall with the help of a reputable clinical research platform.